Unlocking the secret histories of bats in natural history collections
Article by: Caitlin J.Campbell, Graduate Assistant at the UF Department of Biology
Animal migration connects biodiversity with the function and structure of ecosystems at a global scale. Despite having profound and far-reaching effects (affecting the movement of nutrients, energy, and parasites; and altering local- and region-scale biodiversity, and ecosystem and structure and function), these migratory linkages are generally poorly understood (1). Increasingly, the phenomenon of animal migration is threatened by anthropogenic impacts, which include barriers within migration routes, habitat destruction, climate change, and over-exploitation (2, 3). As an additive factor, some migratory species may be more vulnerable to extinction than non-migrating counterparts (4, 5). The knowledge gaps in our understanding of the structure and functions of animal migrations impedes basic scientific understanding as well as the conservation of migratory species and connected ecosystems (6).
Bats, which make up about a quarter of all mammal species, are among the least understood mammals. Although they are essential to the health of many ecosystems—and responsible for consuming huge quantities of crop pests and pollinating many important crops including mangos, bananas, and the agave used in tequila (7–9)— a startlingly large fraction of bat species are in decline (10). Many are migratory, but we really only have loose estimates of how many (11). Many bats are long distance-migrants, including the favorite hoary bat. Studying bat migration is uniquely challenging, since they are very challenging to capture and most long-distance migrants are too small for satellite tracking. Because of the difficulties of studying the movement of individual bats, traditional methods of discovering the wonders of bat migration are missing. Therefore, we must apply other methods.
 |
Examples of migratory bats. Left, lesser long-nosed bat (photo by J. Scott Altenbach), the common noctule (photo), and the hoary bat (photo by Brock Fenton); right, the little collared fruit bat (photo by Natalie Weber), the straw-colored fruit bat (photo by U.S.F.W.S.), and the mexican free-tailed bat (photo by Bureau of Land Management). |
Natural history museum collections may seem an unlikely resource to studying animal migration-- after all, most specimens contained within are long since deceased. However, I apply what could be called “animal CSI”, using chemical signals in animal tissue to estimate where that individual likely grew that tissue. Specifically, I look at stable hydrogen isotopes-- the kind found in all forms of water-- which naturally vary slightly in mass. This slight variation doesn’t affect much, but it does affect the likelihood of a particular water molecule precipitating out of a cloud. This interaction leads to geographic differences in relative abundance of protium and deuterium in the local groundwater. Because animal tissues reflect those of the environment where they were synthesized, one can use stable hydrogen isotope values of (in my case) bat fur to infer the location(s) where the fur was grown. Because fur is an inert tissue, the stable isotope ratios inside do not change over time, and even older specimens can be used to learn a whole host of details about how and where an organism lived.
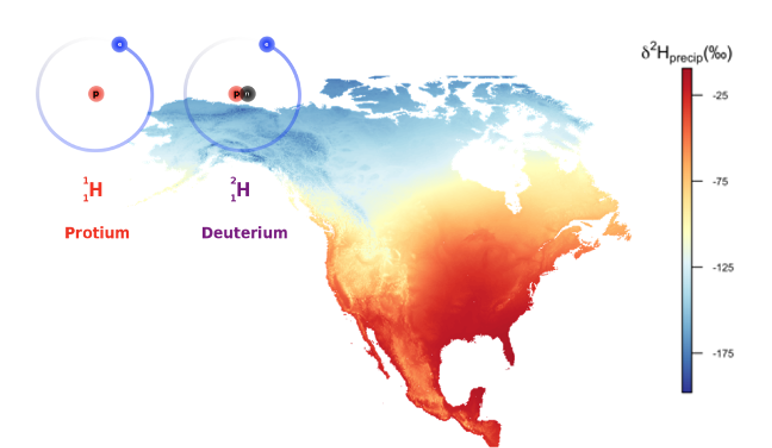 |
Left: The vast majority of hydrogen atoms (99.984%) are protium atoms. A very small amount (0.015%) are deuterium-- a slightly heavier version that is stable and varies geographically in its proportional abundance with respect to protium. Right: A model of stable hydrogen isotope values in North American precipitation. |
The importance of natural history collections is particularly essential when establishing the relationship between environmental conditions and organism’s tissue. I used freshly-grown bat hair, which should closely reflect the local environment where the bat was found, to refine and develop several methods for mapping and comparing the likely origins of hundreds of bats, based on the stable hydrogen isotopes in their fur. These tools can be applied to group bats into those that likely summered together in different regions, and to incorporate known sources of error so models can reflect our confidence in their respective models.
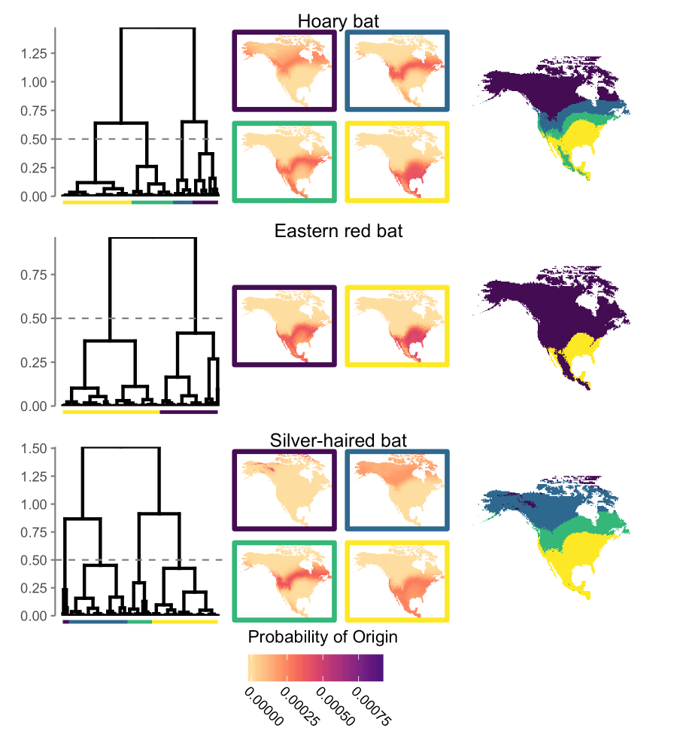 |
Clustering approaches group individuals by similar origin, which increases the aggregated probability-of-origin across groups-- even when very little is known about the potential geographic origins of individual bats. |
I will apply these tools, built on natural history collections, to future study of bat migration and conservation. In the meantime, October is Bat Appreciation Month, and you can learn more about bats and their important role in ecosystems around the world at batcon.org, and celebrate at at batweek.org. I also profiled one of my favorite species of bat, the hoary bat, for iDigBio.
Works cited
- S. Bauer, B. J. Hoye, Migratory animals couple biodiversity and ecosystem functioning worldwide. Science. 344 (2014), doi:10.1126/science.1242552.
- D. S. Wilcove, M. Wikelski, Going, going, gone: Is animal migration disappearing? PLoS Biology. 6, 1361–1364 (2008).
- M. A. Tucker, K. Böhning-Gaese, W. F. Fagan, J. M. Fryxell, B. Van Moorter, S. C. Alberts, A. H. Ali, A. M. Allen, N. Attias, T. Avgar, H. Bartlam-Brooks, B. Bayarbaatar, J. L. Belant, A. Bertassoni, D. Beyer, L. Bidner, F. M. Van Beest, S. Blake, N. Blaum, C. Bracis, D. Brown, P. J. N. De Bruyn, F. Cagnacci, J. M. Calabrese, C. Camilo-Alves, S. Chamaillé-Jammes, A. Chiaradia, S. C. Davidson, T. Dennis, S. DeStefano, D. Diefenbach, I. Douglas-Hamilton, J. Fennessy, C. Fichtel, W. Fiedler, C. Fischer, I. Fischhoff, C. H. Fleming, A. T. Ford, S. A. Fritz, B. Gehr, J. R. Goheen, E. Gurarie, M. Hebblewhite, M. Heurich, A. J. M. Hewison, C. Hof, E. Hurme, L. A. Isbell, R. Janssen, F. Jeltsch, P. Kaczensky, A. Kane, P. M. Kappeler, M. Kauffman, R. Kays, D. Kimuyu, F. Koch, B. Kranstauber, S. LaPoint, P. Leimgruber, J. D. C. Linnell, P. López-López, A. C. Markham, J. Mattisson, E. P. Medici, U. Mellone, E. Merrill, G. De MirandaMourão, R. G. Morato, N. Morellet, T. A. Morrison, S. L. Díaz-Muñoz, A. Mysterud, D. Nandintsetseg, R. Nathan, A. Niamir, J. Odden, R. B. O’Hara, L. G. R. Oliveira-Santos, K. A. Olson, B. D. Patterson, R. C. De Paula, L. Pedrotti, B. Reineking, M. Rimmler, T. L. Rogers, C. M. Rolandsen, C. S. Rosenberry, D. I. Rubenstein, K. Safi, S. Saïd, N. Sapir, H. Sawyer, N. M. Schmidt, N. Selva, A. Sergiel, E. Shiilegdamba, J. P. Silva, N. Singh, E. J. Solberg, O. Spiegel, O. Strand, S. Sundaresan, W. Ullmann, U. Voigt, J. Wall, D. Wattles, M. Wikelski, C. C. Wilmers, J. W. Wilson, G. Wittemyer, F. Ziȩba, T. Zwijacz-Kozica, T. Mueller, Moving in the Anthropocene: Global reductions in terrestrial mammalian movements. Science. 359, 466–469 (2018).
- F. J. Sanderson, P. F. Donald, D. J. Pain, I. J. Burfield, F. P. J. van Bommel, Long-term population declines in Afro-Palearctic migrant birds. Biological Conservation. 131, 93–105 (2006).
- M. Hardesty-Moore, S. Deinet, R. Freeman, G. C. Titcomb, E. M. Dillon, K. Stears, M. Klope, A. Bui, D. Orr, H. S. Young, A. Miller-ter Kuile, L. F. Hughey, D. J. McCauley, Migration in the Anthropocene: how collective navigation, environmental system and taxonomy shape the vulnerability of migratory species. Philosophical Transactions of the Royal Society B: Biological Sciences. 373, 20170017 (2018).
- K. J. Lohmann, Animal migration research takes wing. Current Biology. 28, R952–R955 (2018).
- P. D. Bank, J. Maine, J. Boyles, E. Paraskevopoulos, A. Kamburov, U. Clumps, Agricultural benefits of bats. Proceedings of the National Academy of Sciences. 112, 12223–12224 (2015).
- J. G. Boyles, P. M. Cryan, G. F. McCracken, T. H. Kunz, Economic importance of bats in agriculture. Science. 332, 41–42 (2011).
- T. H. Kunz, E. B. de Torrez, D. Bauer, T. Lobova, T. H. Fleming, K. L. Parkins, J. a. Clark, T. H. Kunz, E. B. de Torrez, D. Bauer, T. Lobova, T. H. Fleming, Ecosystem services provided by bats. Global Ecology and Conservation. 4, 1–38 (2011).
- W. F. Frick, T. Kingston, J. Flanders, A review of the major threats and challenges to global bat conservation. Annals of the New York Academy of Sciences (2019), doi:10.1111/nyas.14045.
- R. Robinson, H. Crick, J. Learmonth, I. Maclean, C. Thomas, F. Bairlein, M. Forchhammer, C. Francis, J. Gill, B. Godley, J. Harwood, G. Hays, B. Huntley, A. Hutson, G. Pierce, M. Rehfisch, D. Sims, B. Santos, T. Sparks, D. Stroud, M. Visser, Travelling through a warming world: climate change and migratory species. Endang. Species. Res. 7, 87–99 (2009).







